Osmosis is the movement of solvent from higher water potential to lower water potential through a semi-permeable membrane until equilibrium state is maintained. Whereas, diffusion is the movement of ions/solute/solvent particles from region of higher concentration to the region of lower concentration until evenly distributed throughout the available spaces.
Semi-permeable membrane is special type of membrane which allows only solvent molecules and selective solute to pass through it. It is also called as differentially permeable membrane.
Difference between osmosis and diffusion
5 Major difference between osmosis and diffusion
| Osmosis | Diffusion |
|---|---|
| It occurs by movement of solvent molecules from higher water potential(i.e dilute solution) to lower water potential solution(i.e concentrate solution) until equilibrium is maintained. | It occurs by movement of solute/ions/solvent from higher concentrated region to lower concentrated region until evenly distributed throughout the available space. |
| It occurs only in liquid medium by the movement of solvent molecules between their liquid solutions. | It occurs in all 3-types of medium i.e solid, liquid & gases by movement of solute/ion as well as solvent molecules. |
| It require biological membrane (e.g semi-permeable membrane). | It does not requires any biological or physical membrane. |
| It is unidirectional and occurs only in one direction. | It is multidirectional and may occurs in all available directions. |
| The solvent molecules move toward higher concentrated solution resulting in increase in quantity. | There is no such markable increase in quantity of solution or to diffusing region as the molecules are distributed in the available space. But, it may increase density to certain extent. |
What is so special about osmosis compared with diffusion?
Osmosis is a special type of diffusion. It is the scattering of only solvent molecules while there is scattering of both solute and solvent in diffusion to maintain concentration difference. Osmosis requires semi-permeable membrane but diffusion does not require any membrane or medium to happen. In osmosis, water or solvent moves from high water potential to low water potential while movement of solute and solvent occurs from high concentrate region to low concentrate region in diffusion.
Similarities between osmosis and diffusion
- Both are passive transport processes which does not require ATPs or energy during the process.
- They both tends to maintain concentration difference between the two solutions or regions.
Examples of Osmosis
Some of the common examples of osmosis are listed below. These 15+ examples are common and can be observed in our daily life.
- Opening and closing of guard cells of stomata
- Absorption of water from soil by roots
- Germination of seeds by absorbing water
- Maintenance of water equilibrium inside cells and tissues
- Brusting and dying of bacterias inside prickles
- Vomiting blood by leeches on sprinkling salt on its body surface
- Swelling of legs and fingers during rainy season due to frequent touch with water etc.
- Examples of diffusion
- Spreading of perfumes odour in whole room while spraying on body
- Spreading a drop of ink throughout the water of the beaker
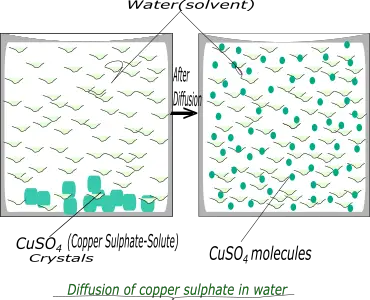
- Dissolving copper sulphate in water
- Dissolving and shaking salt into water
- Diffusion of coal and cigarette smoke in air
- Sweetening of water after putting sugar in it
- Delicious food aroma from kitchen
- Diffusion of oxygen and carbondioxide in lungs etc.